References:
- Maternal & Child Health Nursing: Care of the childbearing & childrearing family, 8th Edition, ISBN 978-1-4963-4813-5, by JoAnne Silbert-Flagg and Adele Pillitteri (Ch. 21)
- Wong’s Nursing Care of Infants and Children, 11th Edition, ISBN 978-0-323-54939-4, by Marilyn J. Hockenberry, David Wilson, and Cheryl C. Rodgers (Ch. 8, pp. 259-283)
- Dr. RPS Maternal and Newborn Care: A Comprehensive Guide and Source Book for Teaching and Learning, 2nd Edition, ISBN 978-971-98-2265-3, by Rosalinda Parado Salustiano (pp. 117-195)
- Comprehensive Nursing Licensure Review Book: Local and International Test Prep, Volume 1, ISBN 978-971-51-3383-8, by Josie Quiambao-Udan
ABO Incompatibilities
Only Rh incompatibilities are often discussed for the topic of isoimmunization/blood incompatibility. However, the rarer but more destructive ABO incompatibilities are a concern when it comes to type O mothers who bear type A or B fetuses. Generally, the same principles of pathophysiology and management apply. I’ve appended a discussion on ABO incompatibilities at the end of this page.
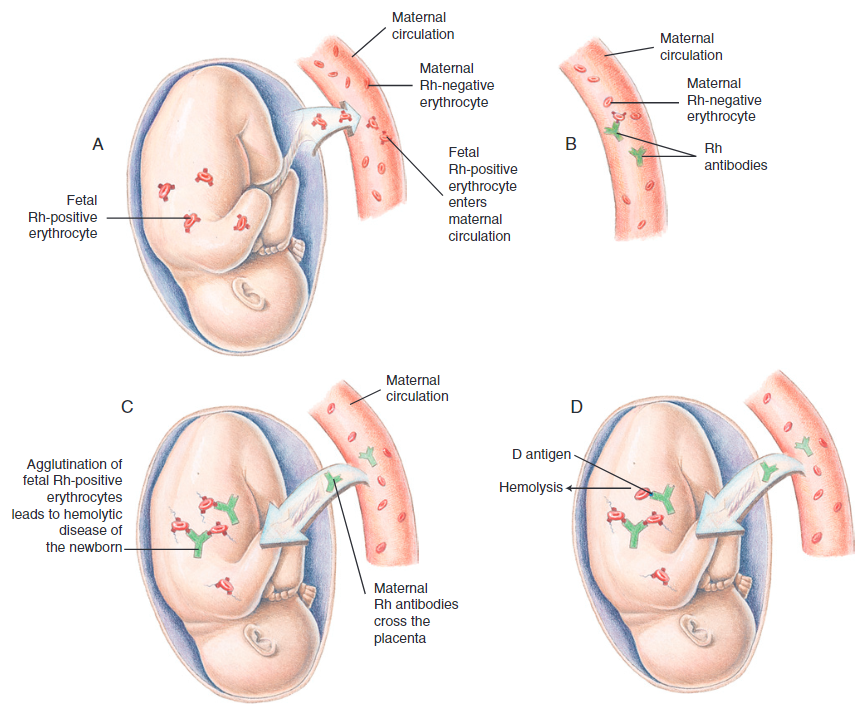
Rh Incompatibility
An Rh incompatibility occurs when an Rh-negative mother carries a fetus with an Rh-positive blood-type. This can occur if the father of the child is either homozygous (DD, 100% of children are Rh positive), or heterogenous (Dd, 50% of the children are Rh positive). The Rh (D) factor is a protein, which means exposure to an Rh-positive fetus will make the mother’s body react in the same manner it would if the invading factor were a substance such as a virus. Antibodies are formed against the invading substance, which are able to cross the placenta and cause hemolysis. In severe red blood cell deficiency, sufficient oxygen transport to body cells cannot be maintained. This condition is terms hemolytic disease of the newborn or erythroblastosis fetalis.
The exchange of required substances between the mother and fetus occurs through means of diffusion and osmosis, transferring oxygen, carbon dioxide, antibodies, proteins, viruses, etc. through the placenta. There is theoretically no connection between fetal blood and maternal blood during pregnancy, but small amounts of fetal blood does enter maternal circulation. Furthermore, procedures such as amniocentesis or percutaneous umbilical blood sampling can allow this to occur. Multiple gestations, abruptio placentae, placenta previa, manual removal of the placenta, and cesarean delivery increase the incidence of transplacental hemorrhage and subsequent isoimmunization. During a first pregnancy, limited exposures will only surmount to a small response. As the placenta separates after birth of the first child, however, there is an active exchange of fetal and maternal blood from damaged villi. This causes most of the maternal antibodies formed against the Rh-positive blood to be formed in the first 72 hours after birth. These become a threat in a second pregnancy.
Assessment
Isoimmunization results in fetal jaundice and an attempt by the fetus to compensate by producing large amounts of immature erythrocytes—erythroblastosis fetalis. In the most severe form of erythroblastosis fetalis, hemolysis results in cardiac decompensation, cardiomegaly, fetal hypoxia, hepatosplenomegaly, ascites and anasarca. Extreme edema occurs, resulting in hydrops fetalis, a pathologic accumulation of at least two or more cavities with a collection of fluid in the fetus.
Neonatal jaundice is not evident as indirect bilirubin is removed by maternal circulation. Pathologic jaundice will occur within 24 hours of life, indicating the occurrence of a hemolytic process. A newborn’s liver is unable to convert the large amounts of indirect bilirubin (fat-soluble—cannot be excreted) into direct bilirubin (water soluble—excreted in urine), leading to jaundice.
A second concern is progressive hypoglycemia, resulting from the use of glucose stores to maintain metabolism in the presence of anemia. A decrease in hemoglobin during the first week of life to a level less than that of the cord blood is a later indication of blood loss or hemolysis.
Fetal Blood Bilirubin Levels
Normally, cord blood has a total serum bilirubin (TsB) level of 0 to 3 mg/dL. This becomes dangerous above 20 mg/dL due to bilirubin-induced neurologic dysfunction (BIND), a wide spectrum of disorders caused by increasing severe hyperbilirubinemia ranging from mild dysfunction to acute bilirubin encephalopathy (ABE).
Diagnosis
- Coombs Test: All women with Rh-negative blood should have an anti-D antibody titer (indirect Coombs test) done at a first pregnancy visit. If the results are normal (0) or the titer is minimal (ratio below 1:8), the test is repeated at week 28 of pregnancy. If this is also normal, no therapy is needed.
- Direct Coombs Testing: fetal blood is used as a sample. This is used to postnatally confirm the presence of antibodies attached to the circulating erythrocytes of affected infants. The Coombs test may be performed on umbilical cord blood samples from infants born to Rh-negative mothers if there is a history of incompatibility or if further investigation is warranted.
- Indirect Coombs Testing: maternal blood is used as a sample. This is more common because maternal blood is easier to obtain. Rising antibody titers arises suspicion of erythroblastosis fetalis in Rh incompatibility. It can also be tested with the optical density of amniotic fluid (how much bilirubin has stained the amniotic fluid).
- Serial Doppler ultrasonography is the gold standard to detect and measure fetal hemoglobin and, subsequently, fetal anemia. If a woman’s anti-D antibody titer is elevated at a first assessment (1:16 or greater), showing Rh sensitization, the well-being of the fetus in this potentially toxic environment will be monitored every 2 weeks (or more often) by Doppler velocity of the fetal middle cerebral artery, a technique that can predict when anemia is present or fetal red cells are being destroyed. If the artery velocity remains high, a fetus is not developing anemia and most likely is an Rh-negative fetus. If the reading is low, it means a fetus is in danger, and immediate birth will be carried out providing the fetus is near term. If the fetus is not near term, efforts to reduce the number of antibodies in the woman or replace damaged red cells in the fetus are begun.
- Amniocentesis can be used to determine fetal blood type for a woman whose antibody screen is positive. A polymerase chain reaction (PCR) can be used to determine fetal blood type, hemoglobin, hematocrit, and presence of maternal antibodies. Chorionic villus sampling has drawbacks that preclude its use, including possible spontaneous abortion of the fetus and fetomaternal hemorrhage. With either method the determination of an Rh-negative fetus requires no further treatment.
- Ultrasonography is an important adjunct that can detect alterations in the placenta, umbilical cord and amniotic fluid, and fetal hydrops with high-resolution ultrasonography. This allows for early, noninvasive treatment before the development of erythroblastosis.
Therapeutic Management
The primary aim of therapeutic management is prevention. Postnatal therapy usually entails phototherapy for mild cases and exchange transfusion for more severe forms. Aggressive interventions are required for hydrops fetalis—pericardial and pleural fluid aspiration, mechanical ventilatory support, and inotrope therapy. The hemolytic process, if untreated, will eventually result in severe anemia between the 7th and 21st day of life.
RhIg Administration
The main method of management is with an intramuscular infusion of RhIG, a commercial preparation of passive Rh (D) antibodies against the Rh factor. These are not able to cross the placenta, as they are not of the IgG type (the only type that crosses the placenta). The infusion eliminates fetal blood that has entered maternal circulation before the body is able to recognize and produce antibodies against the Rh (D) factor. It is given twice in an Rh-negative pregnant client:
- Given at 28 weeks of pregnancy. Its effects last up to three months—12 weeks. It may be given earlier in case of risk of exposure such as bleeding, miscarriage, abortion, invasive procedures, or trauma. If the pregnancy extends beyond its effect, another dose may be given.
- Given within 72 hours after delivery of an Rh-positive baby (or other possible exposures mentioned previously) to prevent antibody formation. Determination of the newborn’s blood type is done by obtaining a sample from cord blood after birth. If the newborn is Rh negative, no antibodies will be formed, making a passive antibody injection unnecessary.
The passive antibodies infused are transient and begin to be destroyed in two weeks to two months. Only a small amount of natural maternal antibodies will remain, ensuring a safe intrauterine environment for any future pregnancies.
IVIG Therapy
Studies have also demonstrated the effectiveness of neonatal IVIG therapy at decreasing the severity of RBC destruction and subsequent development of neonatal jaundice. IVIG is believed to attack the maternal cells that destroy neonatal RBCs, slowing the progression of bilirubin production. This therapy, often used in conjunction with phototherapy, may decrease the necessity for exchange transfusion. Maternal administration of high-dose IVIG, alone or in combination with plasmapheresis, decreases the fetal effects of Rh isoimmunization.
Intrauterine Transfusion
A blood transfusion may be performed for a fetus in utero by injecting red blood cells by amniocentesis technique directly into a vessel in the fetal cord or depositing them in the fetal abdomen (only used if vascular access is impossible; higher associated fetal risks) where they migrate into fetal circulation. The need for this therapy is based on the antenatal diagnosis of fetal anemia by serial Doppler assessments of middle cerebral artery peak systolic velocity.
- Determination of the fetal blood type may be done, or the transfusion may use group O negative blood if the fetal blood type is unknown. The amount ranges from 75 to 150 mL of washed red blood cells depending on the age of the fetus.
- Type O Rh-negative packed RBCs is infused to raise the fetal hematocrit to 40%–50%. Fetal movement is inhibited during transfusion to reduce risk by administering a drug such as vecuronium bromide for temporary fetal paralysis.
- After deposition of the blood, the cannula is withdrawn and the woman is urged to rest for approximately 30 minutes while fetal heart sounds and uterine activity are monitored.
- Another transfusion may be required, as frequent as every two weeks. Once fetal maturity is adequate (mature lecithin/sphingomyelin ratio), birth is induced.
This procedure has its own set of complications. Lacerations or even unintentional induction of labor contractions may occur. However, these are not of greater risk than untreated isoimmunization. After the procedure, RhIG is also given.
After birth, phototherapy may be required due to the excess bilirubin produced by hemolysis. An exchange transfusion may also be performed. Counseling may be provided for contraception if the mother does not want to undergo the strain of another pregnancy.
Bilirubin Management
Bilirubin levels in blood may be measured by either a blood draw (TsB) or by holding a transcutaneous meter against the infant’s skin (transcutaneous bilirubin [TcB]). Measures that may become necessary to reduce the TsB level in an infant affected by blood incompatibility are:
- Early Feeding: bilirubin is removed from the body by being excreted through the feces. Therefore, the sooner bowel elimination begins, the sooner bilirubin removal begins. Feeding with either breastmilk or formula are both effective.
- Phototherapy: exposure of the infant to continuous specialized light (e.g., quartz halogen, cool white daylight, special blue fluorescent light) from 12 to 30 in. above the newborn’s bassinet or incubator accelerates the conversion of unconjugated (fat-soluble) bilirubin into conjugated (water-soluble) bilirubin. Phototherapy is indicated in term infants whose TsB rises to 10–12 mg/dL at 24 hours of age. Preterm infants may begin treatment at lower levels.
Considerations for Phototherapy
- Blindness: Continuous exposure to bright lights by phototherapy may be harmful to a newborn’s retina, so the infant’s eyes must always be covered while under bilirubin lights. Alternatively, bilirubin blankets may also be used. Check the eye covering/mask frequently to be certain it is still in place. Displacement of the cover most often occurs when the infant cries as they wake for a feeding. Urge parents to respond quickly to avoid eye damage or possible suffocation by the mask being pushed down onto the nose. Remove the infant from under the lights regularly for a period of visual stimulation, bonding and feeding.
- Sterility: The infant is undressed except for a diaper to protect the ovaries or testes and so as much skin surface as possible is exposed to the light.
- Dehydration: Phototherapy will increase insensitive water losses. Monitor for dehydration and continue frequent feeding for the infant. Assess skin turgor and intake and output to ensure dehydration is not occurring from the warm environment.
- Overheating: Monitor the infant’s axillary temperature to prevent them from overheating under the bright lights.
- Loose stools: the excretion of bilirubin may stimulate peristalsis, which leads to looser stools. The presence of bilirubin also turns stools a bright green and irritating to the skin.
- Discolored urine: the formation of urobilinogen can turn the urine a dark color.
- Tin-mesoporphyrin, a heme-oxygenase inhibitor, slows down the metabolic processes that produce bilirubin. This allows the newborn’s body systems to manage excretion of bilirubin more readily.
Exchange Transfusion
The necessity of exchange transfusions have greatly been reduced with intensive phototherapy in conjunction with hydration and close monitoring of serum bilirubin levels. This therapy may be used for any condition that leads to hyperbilirubinemia or polycythemia. It reduces the serum concentration of indirect bilirubin and can prevent heart failure in infants with severe anemia or polycythemia.
- A small amount (2–10 mL) of the infant’s blood is drawn and replaced with equal amounts of donor blood. This is repeated multiple times until the desired volume—usually double the infant’s blood volume (~85 ml/kg body weight)—has been exchanged. This replaces approximately 85% of sensitized neonatal blood.
- The transfusion should be done under a radiant heat warmer to maintain body temperature and keep the infant from expending energy. Donor blood must be maintained at room temperature. The donor blood used is O Rh-negative blood.
- Monitor bilirubin levels for 2 or 3 days after the transfusion to ensure the level of indirect bilirubin is not rising again and that no further phototherapy or transfusion is necessary.
Nursing Care Management
The initial nursing responsibility is recognizing the early onset of neonatal jaundice. The possibility of hemolytic disease can be anticipated from the prenatal and perinatal history. Prenatal evidence of incompatibility, maternal blood type O, and a positive Coombs test are cause for increased vigilance for early signs of jaundice in an infant.
- Phototherapy: all considerations mentioned for Phototherapy should be taken into account.
- Exchange Transfusion: the infant is kept on NPO during the procedure. Document blood volumes exchanged, including the amount of blood withdrawn and infused; the time of the procedure; and the cumulative record of the total volume exchanges. Vital signs are also frequently evaluated. The procedure must be stopped if signs of cardiorespiratory function is altered, and only continued once they are stable. The nurse also monitors for signs of transfusion reactions (e.g., temperature instability, hypotension, tachycardia, bradycardia, rash) and maintains adequate neonatal thermoregulation, blood glucose levels, and fluid balance.
- Family Support: parents often feel guilty because they think they cased the incompatibility. Parents should never be made to feel responsible or negligent. The nurse encourages them to express their thoughts. The nurse should praise parents for actions they took to prevent any problems, such as frequent antepartum examinations and blood tests.
ABO Incompatibility
Hemolytic disease can also occur when the major blood group antigens of the fetus are different from those of the mother. The presence or absence of antibodies and antigens determines whether agglutination will occur. Antibodies in the plasma of one blood group (except the AB group, which contains no antibodies) will produce agglutination (clumping) when mixed with antigens of a different blood group. Naturally occurring antibodies in the recipient’s blood cause agglutination of a donor’s RBCs. The agglutinated donor cells become trapped in peripheral blood vessels, where they hemolyze releasing large amounts of bilirubin into the circulation.
The most common blood group incompatibility in the neonate is between a mother with O blood group and an infant with A or B blood group. Unlike the Rh reaction, ABO incompatibility may occur in the first pregnancy. The risk of significant hemolysis in subsequent pregnancies is higher when the first pregnancy is complicated by ABO incompatibility.
Assessment
- Jaundice usually appears during the first 24 hours after birth. Levels of unconjugated bilirubin rise rapidly. Jaundice is usually not present at birth. Hepatosplenomegaly and varying degrees of hydrops may be evident.
- Anemia results from hemolysis of large numbers of erythrocytes.
- Hyperbilirubinemia results from the liver’s inability to conjugate and excrete the excess bilirubin.
- Hypoglycemia may be caused by pancreatic cell hyperplasia originating from elevated glucose levels in-utero from stress due to the hemolytic disease. Hemolytic disease → Increased stress → Signaling for more glucose → Increased insulin requirements → Hypoglycemia once removed from the uterine environment.
Diagnosis and Treatment
The initial diagnosis is often more difficult because the direct Coombs test may be negative or weakly reactive. The presence of jaundice within the first 24 hours, elevated serum bilirubin levels, RBC spherocytosis, and increased erythrocyte production is diagnostic of ABO incompatibility.
- Early detection
- Phototherapy for the reduction of hyperbilirubinemia.
- In some centers IVIG transfusions are used in combination with phototherapy to treat ABO incompatibility.
- Exchange transfusion is not commonly required for ABO incompatibility except when phototherapy fails to decrease bilirubin concentrations.
- The neonatal administration of tin-mesoporphyrin (a heme-oxygenase inhibitor) may also serve to decrease the effects of hemolysis associated with ABO incompatibility.