References:
- Wong’s Nursing Care of Infants and Children, 11th Edition, ISBN 978-0-323-54939-4, by Marilyn J. Hockenberry, David Wilson, and Cheryl C. Rodgers (pp. 1085–1088)
Also Read
Leukemia is a broad term given to a group of malignant diseases of the bone marrow, blood, and lymphatic system. In healthy children, the bone marrow makes blood stem cells that mature to become lymphoid or myeloid stem cells. Myeloid cells differentiate into red blood cells, platelets, and white blood cells. Lymphoid stem cells become lymphoblasts that differentiate into B lymphocytes, T lymphocytes, and natural killer cells. In acute leukemias, immature cells predominate that cannot function effectively. Two types of leukemia are seen most often in children: acute lymphoblastic leukemia (ALL) and acute myeloid leukemia (AML).
Acute Lymphoblastic Leukemia
Previous names for ALL include acute lymphatic, lymphocytic, and lymphoid leukemia. ALL is the most common form of childhood cancer. It is more common in boys than in girls, and in Caucasians more than in African Americans.
- The peak age of onset is 2 to 4 years old.
- Risk factors include prenatal exposure to x-rays, previous treatment with chemotherapy, and certain genetic conditions (e.g., Down syndrome, Bloom syndrome, Fanconi anemia) and various chromosomal abnormalities.
Clinical Manifestations
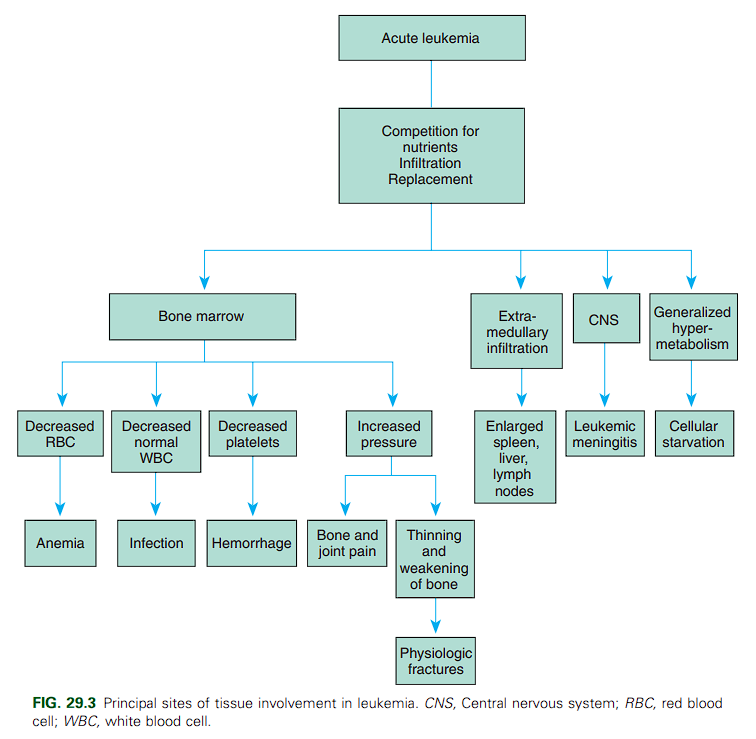
The onset of leukemia varies from acute to insidious. In most instances the child displays remarkably few symptoms. Signs and symptoms of ALL reflect infiltration of the bone marrow by nonfunctional leukemic cells (“blasts”). The three main consequences of bone marrow infiltration are:
- Anemia from decreased erythrocytes
- Infection from neutropenia
- Bleeding from decreased platelet production. Approximately half of patients have an elevated white blood cell count at presentation (>10,000/mm³).
Other signs and symptoms indicate leukemic cell infiltration of other organs; highly vascular organs, such as the spleen and liver, are most severely affected. Commonly seen are hepatosplenomegaly (68% of patients), splenomegaly (63%), fever (61%), lymphadenopathy (50%), bleeding (e.g., petechiae or purpura, 48%), and bone pain (23%).
Diagnostic Evaluation
Leukemia is usually suspected from the history, physical manifestations, and a peripheral blood smear that contains leukemic blasts, frequently in combination with low blood counts.
- Thorough history and physical examination
- Laboratory tests— CBC with differential, blood chemistries
- Bone marrow aspiration/biopsy— cytogenetic analysis, immunophenotyping. The definitive diagnosis is based on analysis of the bone marrow sample. Typically, bone marrow from a child with ALL shows a monotonous infiltrate of blast cells.
- Lumbar Puncture is done after diagnosis to determine the presence of CNS involvement. While uncommon, this is often asymptomatic and warrants screening.
Therapeutic Management
Treatment of ALL is based on risk groups defined by clinical and laboratory findings. Children with a higher risk of relapse/recurrence are treated with the most intensive therapy. Although specifics for various risk groups vary, treatment is generally divided into phases: induction, CNS preventive therapy/consolidation, interim maintenance, delayed intensification, maintenance or continuation therapy, and remission.
- Induction: almost immediately after confirmation of the diagnosis, induction therapy begins and lasts for 4 to 5 weeks. The principal drugs are the corticosteroids (dexamethasone or prednisone), vincristine, and L-asparaginase, with or without an anthracycline.
- A complete remission is determined by the presence of less than 5% blast cells in the bone marrow and no detectable leukemia in extramedullary sites.
- Because many of the drugs also cause myelosuppression of normal blood elements, the period immediately after a remission can be critical. The body is defenseless against invading organisms (especially normal bacterial flora) and susceptible to spontaneous hemorrhage. Consequently, supportive therapy (transfusion, antibacterials, antifungals) during this time is essential.
- CNS preventive therapy is based on the understanding that leukemic cells could be present in the CNS where they are protected from many systemic chemotherapy drugs by the blood-brain barrier. For this reason, all children receive CNS prophylactic therapy. The combination of intrathecal chemotherapy (either methotrexate alone or in combination with cytarabine and hydrocortisone) plus CNS-directed systemic chemotherapy (dexamethasone, L-asparaginase, and high-dose methotrexate with leukovorin rescue) is standard; cranial radiation may be used for children at highest risk for CNS relapse.
- Past clinical trials have shown that postinduction therapy is needed to maintain remission. Treatment regimens vary, but all patients receive consolidation/intensification after achieving a complete remission. Most commonly used is the Berlin, Frankfurt, Münster (BFM) “backbone” (a very intensive chemotherapy regimen from the International BFM Study Group that greatly increased ALL survival). This backbone includes consolidation with cyclophosphamide, cytarabine, and mercaptopurine given initially, followed by interim maintenance with high-dose methotrexate with or without leukovorin rescue.
- Next, delayed intensification is given using drugs and schedules similar to those used in the induction and consolidation phases.
- This is followed by maintenance therapy with daily mercaptopurine and weekly low-dose methotrexate; vincristine and a corticosteroid may be given, as well as continued intrathecal therapy. A critical challenge during maintenance therapy is medication adherence because it has been shown that anything less than 95% adherence increases the risk of relapse.
Acute Myeloid Leukemia
AML accounts for 20% of all cases of childhood leukemia. The incidence is similar for males and females, and higher rates are seen during the first year of life. AML is associated with a variety of predisposition syndromes, including constitutional chromosome abnormalities, inherited single gene mutations, and inherited cytopenias. In addition, therapy-related AML can be caused by treatment with certain chemotherapeutic drugs and/or radiation therapy.
Clinical Manifestations
Many signs and symptoms of AML are similar to those of ALL and reflect infiltration of the bone marrow or extramedullary sites by myeloblasts. Typical signs and symptoms include:
- Fever with or without an infection
- Night sweats
- Shortness of breath, weakness or fatigue
- Easy bruising/bleeding, petechiae
- Bone or joint pain
- Eczema-like skin rash.
- Painless lumps (leukemia cutis) that may be blue or purple in color may appear in the neck, underarm, abdomen, groin, or elsewhere. Other painless lumps, blue-green in color and called chloromas, may be found around the eyes. These are abnormal invasion of tissue by malignant white blood cells.
Diagnostic Evaluation
With a few differences, the workup for AML is similar to that for ALL. Procedures and test include physical examination and history, CBC with differential, blood chemistries, chest x-ray, bone marrow aspiration/biopsy, cytogenetic analysis, RT-PCR test, immunophenotyping, molecular testing, and an LP. A chloroma may be biopsied if present.
Therapeutic Management
Treatment for AML is generally more intense and of shorter duration than treatment for ALL. Chemotherapy is the main treatment modality for AML and is generally given in two phases: induction followed by postremission consolidation/intensification. Maintenance therapy has not been shown to improve outcome and is therefore not used in most treatment regimens.
- Standard remission induction consists of a three-drug regimen of cytarabine, daunorubin, and etoposide. CNS treatment with intrathecal medication is usually included in treatment regimens; CNS irradiation is not usually used in AML treatment. Once remission is achieved (no signs of leukemia detected), stem-cell transplant (SCT) may be performed. Alternatively, intensive chemotherapy may be given and SCT reserved in case the AML relapses.
- Intensification chemotherapy usually consists of high-dose cytarabine with or without daunorubicin; intrathecal chemotherapy is given every 1 to 2 months during intensification.
Nursing Care Management
Nursing care of the child with acute leukemia, ALL or AML, is directly related to the regimen of therapy. Myelosuppression, drug toxicity, and leukemic infiltration cause secondary complications that necessitate supportive physical care.
- Prepare the Family for Diagnostic and Therapeutic Procedures: both the children and their parents should be provided with explanations, rationale, what can be expected, and what they can do to help during procedures.
- Provide Continued Emotional Support