References:
- Brunner & Suddarth’s Textbook of Medical-Surgical Nursing, 15th Edition, ISBN 978-197-51-6103-3, by Janice L. Hinkle, Kerry H. Cheever, and Kristen J. Overbaugh (Ch. 30, [ebook] pp. 2609–2638)
Also Read
The term leukocytosis refers to an increase of leukocytes (WBCs) in the circulation. Typically, only one specific cell type is increased. Because the proportions of several types of leukocytes (e.g., eosinophils, basophils, monocytes) are small, an increase in other types can be great enough to elevate the total leukocyte count, particularly the neutrophils or lymphocytes. Although leukocytosis can be a normal response to increased need (e.g., in acute infection), the elevation in leukocytes should decrease as the physiologic need decreases. A prolonged or progressively increasing elevation in leukocytes is abnormal and should be evaluated. A significant cause of persistent leukocytosis is a hematologic malignancy (i.e., leukemia).
The common feature of the leukemias is an unregulated proliferation of leukocytes in the bone marrow.
- In acute forms (or late stages of chronic forms), the proliferation of leukemic cells leaves little room for normal cell production. There can also be a proliferation of cells in the liver and spleen (extramedullary hematopoiesis). With acute forms, there can be infiltration of leukemic cells in other organs, such as the meninges, lymph nodes, gums, and skin.
- The cause of leukemia is not fully known, but exposure to radiation or chemicals, certain genetic disorders, and viral infections are known to be risk factors for certain types of leukemia. Bone marrow damage from pelvic radiation or certain types of chemotherapy drugs can cause acute leukemia, typically occurring years after treatment for another malignancy.
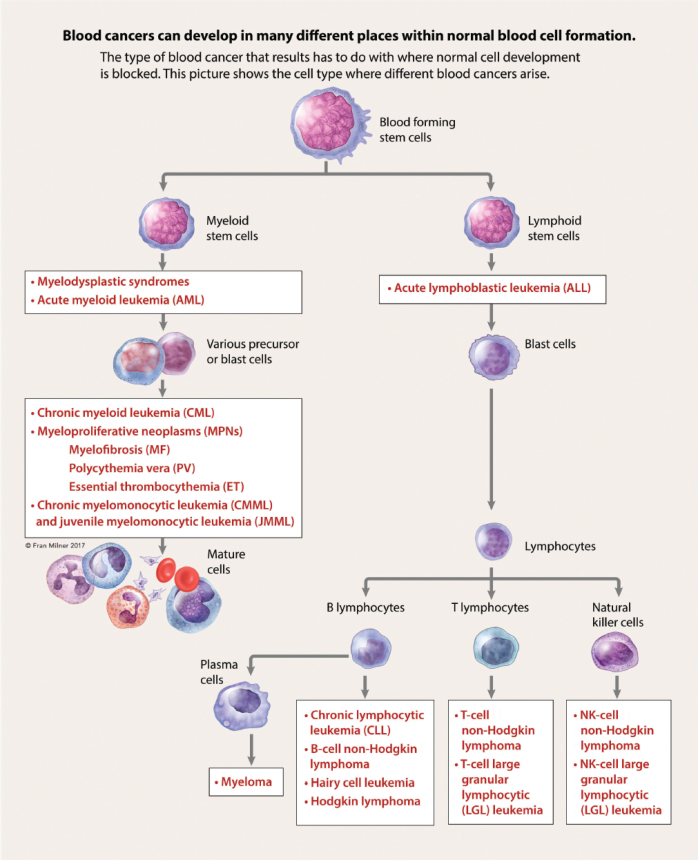
Types of Leukemia
The leukemias are commonly classified according to the stem cell line involved, either:
- Lymphoid: referring to stem cells that produce lymphocytes, or
- Myeloid: referring to stem cells that produce nonlymphoid blood cells.
They are also classified as either acute or chronic , based on the time it takes for symptoms to evolve and the phase of cell development that is halted (i.e., with few leukocytes differentiating beyond that phase):
- Acute leukemia features an abrupt onset of symptoms, often within a few weeks. Leukocyte development halts at the blast phase, and thus most leukocytes are undifferentiated cells or blasts. Acute leukemia can progress rapidly, with death occurring within weeks to months without aggressive treatment.
- Chronic leukemia features symptoms that evolve over a period of months to years, and the majority of leukocytes produced are mature. This progresses more slowly, with the disease trajectory potentially extending for years.
Acute Myeloid Leukemia
Acute myeloid leukemia (AML) originates due to a series of genetic mutations in the myeloid hematopoietic stem cells leading to clonal development of abnormal blast cells. As these blast cells (i.e., immature leukocytes) continue to proliferate, they crowd out normal bone marrow production resulting in anemia, thrombocytopenia, and either low or elevated WBC counts. It may rarely be normal. There is also impaired development of all myeloid cells: monocytes, granulocytes (i.e., neutrophils, basophils, eosinophils), erythrocytes, and platelets.
- AML is the most common form of leukemia, as well as the most common cause of death from all leukemias. It affects any age groups, increasing in incidence with age. The median age of diagnosis is 68 (Leukemia and Lymphoma Society, 2019). Leukemia accounts for 1% of annual cancer-related deaths.
- The exact cause of AML is unclear, but risk factors include:
- Increasing age
- Males
- Exposure to chemicals (e.g., benzene, pesticides)
- Exposure to ionizing radiation
- A history of prior treatment with chemotherapeutic drugs (e.g., alkylating agents, topoisomerase inhibitors)
- Tobacco smoking
- Other blood disorders (e.g., myeloproliferative diseases)
- Several genetic disorders (e.g., Down syndrome, Trisomy 8, or Fanconi anemia)
Prognosis
Prognosis and survival rates are highly variable.
- Better prognosis; more favorable: younger age at diagnosis, more favorable cytogenetic alteration (also associated with younger age), and few concurrent (or mild) health problems.
- Poorer prognosis; less favorable: patients with significant comorbidities, of older age, with cytogenetic features deemed to be adverse, or who are frail, are more likely to have a poor prognosis. AML evolving from a preexisting clonal myeloid disease or from prior cytotoxic therapy for another malignancy or immune disease.
Clinical Manifestations
AML often presents, initially, as nonspecific complaints that can abruptly occur or gradually worsen over time. The signs and symptoms result from inadequate production of normal blood cells, especially as the leukemic cells increasingly crowd out the bone marrow.
- Neutropenia results in fever and infection. However, fevers may not be related to infection.
- Anemia results in pallor, fatigue, weakness, dyspnea on exertion, and dizziness.
- Thrombocytopenia results in ecchymoses, petechiae, epistaxis, and gingival bleeding.
The proliferation of leukemic cells within organs results in a variety of additional symptoms:
- Pain from an enlarged liver or spleen.
- Hyperplasia of the gums.
- Bone pain from expansion of marrow.
- Petechiae or ecchymoses are common on the skin; occasionally, leukemic infiltrates (leukemia cutis) are also seen.
- Gingiva or synovial spaces can also be infiltrated.
- Lymphadenopathy and splenomegaly is rare.
Assessment and Diagnostic Findings
- Complete blood count commonly shows a decrease in both erythrocytes and platelets. Although the total leukocyte count can be low, normal, or high, the percentage of normal cells is usually vastly decreased.
- A bone marrow analysis shows an excess (more than 20%) of blast cells; this is the hallmark of the diagnosis.
- Further classification of AML into seven subgroups can be done based on cytogenetics, histology, and morphology of the blasts, as well as the presence of genetic mutations.
- While prognosis vary between subtypes, the clinical course and treatment differ substantially with only one subtype: acute promyelocytic leukemia (APL, or AML-M3). This type has a higher potential for fatal coagulopathies; however, the potential to cure this form of AML is high.
Medical Management
The overall goal is complete remission of the disease. For this, chemotherapy treatment is administered in two parts:
- Induction therapy: high doses of cytarabine and either daunorubicin, idarubicin, or mitoxantrone; etoposide is occasionally added to the regimen.
- Lower-intensity therapy is used for older patients who cannot tolerate the standard regimen.
Care of Patients with Chemotherapy
During therapy, chemotherapy not only destroys leukemic cells but also healthy cells, requiring patients to be hospitalized for several weeks (typically 4 to 6 weeks) due to severe and potentially life-threatening side effects, such as neutropenia. In some patients, an absolute neutrophil count of zero is not uncommon. Anemia, and severe thrombocytopenia is also common.
During this time, the patient is typically very ill, with bacterial, fungal, and occasionally viral infections; bleeding; and severe mucositis, which cause pain, diarrhea, and an inability to maintain adequate nutrition.
Management consists of administering blood products and promptly treating infections. For patients who have a life-threatening infection, granulocytic growth factor may be used during the induction phase to shorten the neutropenic phase. The patient is discharged when blood counts are normal and infection risk is diminished.
- Consolidation therapy is began when the patient has recovered from the induction therapy. It aims to eliminate any residual leukemic cells that are not clinically detectable and to reduce the chance for recurrence of leukemia. Usually, this includes one cycle of the same regimen used in induction therapy with lower dosages. Multiple treatment cycles of various agents are used, usually containing some form of cytarabine.
Allogeneic stem cell transplant is the most common form of hematopoietic stem cell transplant (HSCT) used in the treatment of AML. It is routinely done following induction and consolidation therapy.
- In severe cases, HSCT may begin after induction.
- The process of HSCT requires that patients begin by receiving high-dose, aggressive chemotherapy, sometimes in tandem with radiation therapy, to destroy the hematopoietic functioning in the bone marrow and to kill any residual leukemic cells. This is called conditioning therapy. Then, the patient is given human leukocyte antigen (HLA-matched) donor stem cells via IV infusion to reestablish bone marrow functioning and to create a new immune system.
- The best timing of HSCT remain unclear. Patients with poorer prognosis may benefit from early HSCT, and patients with a good prognosis may not ever require HSCT.
10% to 40% of patients do not achieve complete remission following induction therapy.
- The induction regimen may be repeated until remission or relapse occurs.
- Other approaches include treatment with enasidenib, a cytarabine-based regimen, along with other agents (e.g., cladribine, fludarabine, mitoxantrone, etoposide); palliative care; or use of a hypomethylating agent (e.g., azacitidine or decitabine).
Complications
Bleeding and infection are the major causes of death.
- Bleeding is correlated with the level and duration of platelet deficiency. Ecchymoses and petechiae may appear.
- Major hemorrhages may develop when the platelet count drops to less than 10,000/mm³. The most common bleeding sources include gastrointestinal (GI), pulmonary, vaginal, and intracranial.
- Fever and infection also increases the likelihood of bleeding, for undetermined reasons.
- Disseminated intravascular coagulation is common, particularly in patients with the APL subtype.
- Infection is due to the lack of mature and normal granulocytes that help fight infection. The likelihood of infection increases with the degree and duration of neutropenia.
- Neutrophil counts that persist at less than 100/mm³ dramatically increase the risk of systemic infections.
- Risk for fungal infection also increases. Fungal infections remain difficult to treat; at times, patients may begin prophylactic treatment with antifungal agents.
- Granulocytic growth factors are used to stimulate the bone marrow to produce leukocytes, thereby shortening the period of neutropenia. However, this is not used for patients with APL as it may result in 1differentiation syndrome.
- Fluid and electrolyte imbalance: Massive leukemic cell destruction from chemotherapy results in the release of intracellular electrolytes and fluids into the systemic circulation.
- Increases in uric acid levels, potassium, and phosphate are seen; this process is referred to as tumor lysis syndrome. The increased uric acid and phosphorus levels make the patient vulnerable to renal stone formation and renal colic, which can progress to acute kidney injury.
- Hyperkalemia and hypocalcemia can lead to cardiac arrhythmias; hypotension; neuromuscular effects such as muscle cramps, weakness, and spasm/tetany; and confusion; and seizures may also develop.
- Patients require a high fluid intake, and prophylaxis with allopurinol or rasburicase to prevent crystallization of uric acid and subsequent stone formation.
- GI problems may result from the infiltration of abnormal leukocytes into the abdominal organs and from the toxicity of the chemotherapeutic agents. Anorexia, nausea, vomiting, diarrhea, and severe mucositis are common.
Nursing Management
Nursing management of the patient with acute leukemia, including AML, is discussed later in this page.
Chronic Myeloid Leukemia
Chronic myeloid leukemia (CML) arises from a mutation in the myeloid stem cell. Normal myeloid cells continue to be produced, but there is a pathologic increase in the production of forms of blast cells. Therefore, a wide spectrum of cell types exists within the blood, from blast forms to mature neutrophils.
- Because there is an uncontrolled proliferation of cells, the marrow expands into the cavities of long bones, such as the femur, and cells are also formed in the liver and spleen (extramedullary hematopoiesis), resulting in enlargement of these organs that is sometimes painful.
- CML accounts for 15% of all new cases of leukemia. The average age of a patient at time of diagnosis is 67 years, but CML can occur at any age.
- Risk factors include increasing age, being male, having a history of smoking, and being exposed to high doses of radiation (e.g., atomic bomb survivors).
The Philadelphia Chromosome
CML results from a chromosomal translocation, where a section of deoxyribonucleic acid (DNA) is shifted from chromosome 22 to chromosome 9, forming what is known as a “fusion gene” that is abnormal. The specific fused gene found in all patients with CML is the BCR-ABL gene, which occurs when the BCR gene from chromosome 22 switches places with the ABL gene from chromosome 9.
Normally, the ABL gene signals the cells when to make tyrosine kinase; however, the abnormal BCR-ABL gene (known as the Philadelphia chromosome) signals cells to produce too many leukocytes and is responsible for converting normal cells into leukemic cells.
Clinical Manifestations
There are three stages of CML that present different clinical pictures:
- Chronic Phase: patients have few symptoms—leukocytosis is detected by a CBC performed for some other reason, and complications from the disease itself are rare.
- Accelerated Phase: either insidious or rapid, this marks the process of evolution to the acute form of leukemia (blast crisis). In the accelerated phase of disease, blood counts begin to worsen, new chromosomal changes may be seen on analysis, and symptoms consistent with leukemia may start to appear, such as fatigue, anemia, splenomegaly, or dyspnea. The patient may complain of bone pain and may report fevers (without any obvious sign of infection) and weight loss.
- Blast Crisis: the most advanced phase, where signs and symptoms become like those in AML. Some patients present with leukocytosis, with the WBC count exceeding 100,000/mm³.
- Patients with extremely high leukocyte counts may be dyspneic or slightly confused because of decreased perfusion to the lungs and brain from leukostasis (inhibited blood flow through the capillaries).
- The patient may have an enlarged, tender spleen, and occasionally the liver may also be enlarged and tender.
- Some patients have insidious symptoms, such as malaise, anorexia, and weight loss.
- Lymphadenopathy is uncommon, but if present, indicates late disease and a poor prognosis.
Medical Management
The goal of treatment for CML is to control the disease, either by obtaining remission or by keeping the patient in the chronic phase for as long as possible. CML is not considered to be curable among older adults.
- Tyrosine kinase inhibitors inhibit the bioproduct that signals the body to produce leukocytes; it blocks the signals within the leukemic cells that express the BCR-ABL protein. This prevents a series of chemical reactions that cause the cells to grow and divide, thus inducing complete remission at the cellular level.
- TKI imatinib mesylate is considered to be the standard of care for patients with CML. Newer alternatives (dasatinib, nilotinib) are also approved for first-line therapy. Second-line therapy may include bosutinib and ponatinib.
- Each type of TKI brings a different toxicity profile. These include myelosuppression, pleural effusion, prolonged QT interval, cardiotoxicity, and drug-to-drug interactions (All TKIs are metabolized via cytochrome P450 pathway).
- Drugs that counteract the effects of TKI are corticosteroids, antiseizure medications, antacids, and St. John’s wort.
- Drugs that may increase the effects of TKI include grapefruit juice, certain antibiotics (e.g., clarithromycin), and azole antifungals (e.g., clotrimazole, ketoconazole).
- Allogeneic Hematopoietic Stem Cell Transplant can potentially cure CML in otherwise healthy patients who are younger than 65 years. However, with the development of TKIs, the timing of transplant has come into question. Approximately 90% of patients who receive allogeneic HSCT during the chronic phase of CML are disease free for 5 years or more.
- Induction therapy may be used for patients in blast crisis, as it resembles AML. This uses the same medications for AML or ALL (discussed later). A return to the chronic phase is possible if CML evolves into ALL, but is unlikely if CML evolves into AML. Life-threatening infections and bleeding occur frequently in this phase.
Nursing Management
Most TKIs are oral agents; therefore, their effectiveness is dependent upon the patient’s ability and motivation to adhere to the prescribed treatment regimen. Studies show that adherence rates is only around 50%. In outpatients, studies shows reasons for nonadherence were most commonly due to forgetting to take the drug or delays of 2 or more hours from the prescribed time. Other reported reasons ranged from patient forgetfulness to perceptions of inadequate support.
These drugs may cause side effects that the patient may find difficult to manage, such as fatigue, asthenia (weakness), pruritus (itching), headache, skin rash, and oropharyngeal pain.
- It is extremely important for the nurse to educate the patient about the medication regimen, how to manage side effects, drug interactions, and safe handling.
- The nurse should also monitor the patient for adverse signs and symptoms of therapy, such as decreased urinary output, changes in the electrocardiogram (TKIs can cause arrhythmias and prolonged QT intervals), and myelosuppression (e.g., fevers, chills, changes in the CBC).
Acute Lymphocytic Leukemia
ALL results from an uncontrolled proliferation of immature cells (lymphoblasts) derived from the lymphoid stem cell. The cell of origin is the precursor to the B lymphocyte in approximately 75% of ALL cases; T-lymphocyte ALL occurs in approximately 25% of cases.
- 75% to 80% of all ALL cases are found in children, but can occur at any age. The median age of diagnosis is 15, peaking at 4 years. After 15, it is relatively uncommon, until incidence rises again at 45.
- Risk factors: male gender, older age (especially over 70), prior exposure to chemotherapy or radiation therapy, having certain genetic conditions (e.g., especially Down syndrome; also neurofibromatosis, Klinefelter syndrome, and Fanconi anemia).
Clinical Manifestations
For some patients with ALL, the clinical manifestations may be nonspecific while others have no symptoms initially. The disease is commonly found incidentally with routine laboratory studies or physical exam for another condition.
- At the time of diagnosis, the leukocyte counts may be either higher or lower than normal, with a high proportion of immature lymphoblasts.
- Immature lymphocytes proliferate in the marrow and impede the development of normal myeloid cells. As a result, normal hematopoiesis is inhibited, resulting in reduced numbers of granulocytes, erythrocytes, and platelets.
- Manifestations of leukemic cell infiltration into other organs are more common with ALL than with other forms of leukemia and include pain from an enlarged liver or spleen as well as bone pain. The central nervous system (CNS) is frequently a site for leukemic cells; thus, patients may exhibit cranial nerve palsies or headache and vomiting because of meningeal involvement. Other extranodal sites include the testes and breasts.
Medical Management
The goal of treatment is to obtain remission without excess toxicity and with a rapid hematologic recovery so that additional therapy can be given if needed. Because of the heterogeneity of the disease, treatment plans are based on genetic markers of the disease as well as risk factors of the patient, primarily age. Similar to treatment for AML, treatment for ALL can be grouped into induction, consolidation, and maintenance phases. Because ALL frequently invades the CNS, preventive intrathecal chemotherapy or, less frequently, cranial irradiation, are also a key part of the treatment plan.
Treatment protocols are complex, with a variety of agents and complicated administration schedules. The expected outcome is complete remission. Despite its complexity, treatment can be provided in the outpatient setting in some circumstances until severe complications develop.
- TKIs (e.g., imatinib) are effective in patients with Philadelphia chromosome-positive ALL; these drugs can be used alone or in combination with conventional chemotherapy.
- Lymphoid blast cells are typically very sensitive to corticosteroids and to vinca alkaloids; therefore, these medications are an integral part of the initial induction therapy.
- The corticosteroid dexamethasone is preferred to prednisone, as it is more toxic to lymphoid cells and has better CNS penetration.
- Typically, an anthracycline is included, sometimes with asparaginase.
Once a patient is in remission, special testing (immunophenotyping, immunoglobulin gene rearrangements, T-cell receptor genes, molecular testing) is done to look for residual leukemic cells; these tests can detect as few as a single ALL cell among 10,000 to 100,000 normal cells.
- This minimal residual disease testing is useful as a prognostic indicator. Based on these results and the rapidity in which remission is achieved, a consolidation regimen ensues, using different combinations and dosages of the drugs used in induction therapy; the goal of consolidation is to improve outcomes in those patients at high risk for relapse.
Patients with ALL can experience some unique adverse effects from treatment.
- The use of corticosteroids to treat ALL increases the patient’s susceptibility to infection; viral infections are common.
- Avascular necrosis can occur in patients treated with corticosteroid-based chemotherapy, as well as with transplantation.
- Patients treated with asparaginase are at increased risk for thrombosis. Hepatic toxicity is also common and may necessitate cessation of supportive drugs, such as proton pump inhibitors and certain antibacterial and antifungal drugs.
Allogeneic HSCT may be considered during initial remission if disease features and testing suggest the risk of relapse is high. The development of chimeric antigen receptor (CAR) T cells has significantly improved treatment outcomes and overall survival in patients with ALL. CAR-T therapy utilizes the patient’s own immune system to fight disease; the patient’s own T cells are collected, modified, and reinfused back into the patient.
- Treatment with CAR-T can serve as a bridge prior to transplant and has qualified patients for HSCT who were formerly ineligible. In the context of average-risk disease, HSCT may be postponed until the time of relapse, should it occur. HSCT can improve long-term disease-free survival; however, this potential benefit must be weighed with the risks associated with the procedure, including death and long-term morbid complications.
Nursing Management
Nursing management of the patient with acute leukemia, including ALL, is discussed later in this page.
Chronic Lymphocytic Leukemia
Chronic lymphocytic leukemia (CLL) is a common malignancy of older adults, and the most prevalent type of adult leukemia in the Western world (NCCN, 2019d).
- The average age at diagnosis is 71 years (Leukemia & Lymphoma Society, 2019c).
- CLL is rarely seen in Native Americans and infrequently among people of Asian descent. Unlike other forms of leukemia, a strong familial predisposition exists with CLL; the disease can occur in 10% of those with a first- or second-degree relative with the same diagnosis.
- While many patients will have a normal life expectancy, others will have a very short life expectancy due to the aggressive nature of the disease.
Pathophysiology
CLL is typically derived from a malignant clone of B lymphocytes. A clone proliferates from a cell of origin so that descendent cells are identical to the cell of origin. In contrast to the acute forms of leukemia, most leukemic cells in CLL are fully mature. One possible mechanism that explains this oncogenesis is that these cells can escape apoptosis (programmed cell death), resulting in an excessive accumulation of the cells in the marrow and circulation.
- CLL is characterized by the progressive accumulation of leukemic cells in the bone marrow, blood, and lymphoid tissues.
- Because the lymphocytes are small, they can easily travel through the small capillaries within the circulation, and the pulmonary and cerebral complications of leukocytosis seen with myeloid leukemias are not typically found in CLL.
- However, these cells often accumulate within the lymph nodes and spleen. When it takes less than 12 months for the absolute number of lymphocytes to double (lymphocyte doubling time), a more aggressive disease course may ensue.
Immunophenotyping of the circulating B cells is critical to establish the diagnosis by identifying the presence of a malignant clone of these cells; it is also used to gauge the prognosis (NCCN, 2019d). Other special cytogenetic and molecular analyses (e.g., fluorescence in situ hybridization [FISH]) are also used to guide prognosis and therapy. Beta-2 microglobulin, a protein found on the surface of lymphocytes, can be measured in the serum; an elevated level correlates with a more advanced clinical stage and poorer prognosis.
Autoimmune complications can occur at any stage, as either autoimmune hemolytic anemia or idiopathic thrombocytopenic purpura. In the autoimmune process, the reticuloendothelial system destroys the body’s own erythrocytes or platelets. Patients with CLL also have a greater risk for developing other cancers. Approximately 2% to 10% of patients with CLL will experience transformation of their disease to a very aggressive lymphoma, known as Richter’s transformation (NCCN, 2019d); this transformation results in markedly increased lymphadenopathy, splenomegaly, B symptoms, and survival of only a few months despite treatment. Second cancers typically involve the skin, colon, lung, breast, prostate, and kidney.
B Symptoms
B symptoms are multisystemic symptoms that can manifest with many types of hematopoietic malignancies, including CLL, Hodgkin lymphoma, and non-Hodgkin lymphomas. It is associated with the worst prognoses in patients with CLL, Hodgkin lymphoma and NHL than in patients who do not report B symptoms. These include:
- Fever of at least 100.4°F (38°C) that may come and go over several weeks that is not explained by an underlying infection
- Drenching night sweats
- Unintentional loss of at least 10% body weight over the past 6 months
Clinical Manifestations
Many patients are asymptomatic and are diagnosed incidentally during routine physical examinations or during treatment for another disease.
- Lymphocytosis (an increased lymphocyte) is always present.
- The erythrocyte and platelet counts may be normal or, in later stages of the illness, decreased.
- Lymphadenopathy is common; this can be severe and sometimes painful.
- Splenomegaly may also occur.
- Patients with CLL may also develop B symptoms (see info box above) which portends a worsening prognosis
- T-cell function is impaired and may be the cause of tumor progression and increased susceptibility to second malignancies and infections.
- Life-threatening infections are particularly common with advanced disease, and account for over half of all deaths in this patient population. Viral infections, such as herpes zoster, can become widely disseminated.
- Defects in the complement system are also seen, which results in increased risk of developing infection with encapsulated organisms (e.g., Haemophilus influenzae).
Patients should receive an annual comprehensive skin examination due to a high incidence in skin cancer, and screening guidelines for other cancers (breast, colorectal, lung, prostate) should be followed.
Medical Management
Treatment is begin when the progression of disease is noted. The traditional “watch-and-wait” approach is used for patients with no symptoms at the time of diagnosis. Newer therapies, however, may allow treatment to be initiated sooner in the illness trajectory. Patients with good general health and are likely to tolerate aggressive therapy often achieve a lasting complete remission. In contrast, therapy in those with impaired physical status focuses on controlling bothersome symptoms (e.g., drenching night sweats, painful lymphadenopathy).
- Treatment is variable and can consist of a single immunotherapy agent administered in combination with chemotherapeutic agents, such as an immunotherapeutic antibody against the B-lymphocyte antigen CD20 (e.g., rituximab, ofatumumab, obinutuzumab) with chemotherapeutic agents (e.g., fludarabine, cyclophosphamide, bendamustine, chlorambucil) as initial therapy.
- Fludarabin’s major side effect is prolonged bone marrow suppression, manifested by prolonged periods of neuropenia, lymphopenia, and thombocytopenia, which puts patients at risk for such infections as Pneumocystic jiroveci, Listeria, mycobateria, herpes viruses, and cytomegalobirus (CMV).
- The monoclonal antibody (MoAb) alemtuzumab is often used in combination with other chemotherapeutic agents when the disease is refractory to fludarabine, the patient has very poor prognostic markers, or it is necessary to eradicate residual disease after initial treatment.
- Alemtuzumab targets CD52, an antigen commonly found on CLL cells, allowing it to clear the marrow and circulation of these cells without affecting stem cells. However, CD52 is present on both B and T lymphocytes, predisposing the patient to infection. Prophylactic use of antiviral and antibiotic agents is important and needs to continue for several months after treatment ends.
- When the disease is accompanied by a deletion of or mutation of the TP53 gene, TKIs such as ibrutinib or idelalisib may be used as either monotherapy or in combination with other agents.
- Patients receiving idelalisib or duvelisib have an increased risk of hepatotoxicity, severe diarrhea, colitis, and pneumonitis. TKIs have been found to increase the risk of cardiovascular toxicities, including hypertension, prolonged QT interval, left ventricular dysfunction, and heart failure.
- Due to the high median age at diagnosis, allogeneic HSCT may not be an option, particularly if significant comorbidities exist. However, it may be considered for patients with TP53 deletions or mutations who otherwise have a poor prognosis. This treatment modality may be reserved for those patients with high-risk disease, younger age, and high degree of match from the donor.
Nursing Management
Virtually all patients with CLL have reduced levels of immunoglobulins, and bacterial infections are common, independent of treatment.
- IV treatment with immunoglobulin (IVIG) may be given to select patients with recurrent infection. While studies have not demonstrated improved survival, the rate of developing major infections is reduced.
- Patients with CLL should receive both pneumonia and flu vaccinations as indicated. Live vaccines should be avoided.
- The patient with CLL is at increased risk of a host of infections; nursing interventions focuses on diminishing these risks.
Nursing Management for Acute Leukemia
Assessment
Although the clinical picture varies with the type of leukemia as well as with the treatment implemented, the health history may reveal a range of subtle symptoms reported by the patient before the problem is detectable on physical examination.
- If the patient is hospitalized, assessments should be performed daily, or more frequently as warranted. Because the physical findings may be subtle initially, a thorough, systematic assessment incorporating all body systems is essential.
- The nurse also must closely monitor the results of laboratory studies, including tracking the leukocyte count, ANC, hematocrit, platelet, creatinine and electrolyte levels, and coagulation and hepatic function tests. Culture results need to be reported immediately so that appropriate antimicrobial therapy can begin or be modified.
Diagnosis
Nursing Diagnoses
Based on the assessment data, major nursing diagnoses may include:
- Risk for infection, haemorrhaging, or both
- Impaired oral mucous membrane integrity due to changes in epithelial lining of the GI tract from chemotherapy or prolonged use of antimicrobial medications
- Impaired nutritional status associated with hypermetabolic state, anorexia, mucositis, pain, and nausea
- Acute pain associated with mucositis, leukocyte infiltration of systemic tissues, fever, and infection
- Fatigue and activity intolerance associated with anemia, infection, inadequate nutrition, and deconditioning
- Fluid imbalance associated with renal dysfunction, diarrhea, bleeding, infection, increased metabolic rate, hypoproteinemia, and need for multiple intravenous medications and blood products
- Impaired ability to perform hygiene, impaired ability to dress, and impaired self toileting due to fatigue and malaise
- Anxiety and grief due to uncertainty about future, anticipatory loss, and altered role functioning
- Risk for spiritual distress
- Lack of knowledge about disease process, treatment, complication management, and self-care measures
Complications
Potential complications may include the following:
- Infection
- Bleeding/DIC
- Renal dysfunction
- Cardiac toxicity
- Infertility
- Tumor lysis syndrome
Planning and Goals
The major goals for the patient may include absence of complications and pain, attainment and maintenance of adequate nutrition, activity tolerance, ability to provide self-care and to cope with the diagnosis and prognosis, positive body image, and an understanding of the disease process and its treatment.
Nursing Interventions
- Preventing or managing infection and bleeding:
- Managing mucositis: Although emphasis is placed on the oral mucosa, the entire GI mucosa can be altered, not only by the effects of chemotherapy but also from prolonged administration of antibiotics.
- Improving nutritional intake: The disease process can increase the patient’s metabolic rate and nutritional requirements. Nutritional intake is often reduced because of pain and discomfort associated with stomatitis.
- Encouraging or providing mouth care before and after meals and administering analgesic agents before eating can help increase intake.
- If oral anesthetic agents are used, the patient must be warned to chew with extreme care to avoid inadvertently biting the tongue or buccal mucosa.
- Nausea should not interfere with nutritional intake, because appropriate antiemetic therapy is highly effective. However, nausea can result from antimicrobial therapy, so some antiemetic therapy may still be required after the chemotherapy has been completed.
- Small, frequent feedings of foods that are soft in texture and moderate in temperature may be better tolerated.
- Low-microbial diets may be prescribed (avoiding uncooked fruits or vegetables and those without a peelable skin), although there is little evidence to support this intervention.
- Nutritional supplements are frequently used.
- Daily body weight (as well as intake and output measurements) is useful in monitoring fluid status. Both calorie counts and more formal nutritional assessments are often useful.
- Parenteral nutrition may be required to maintain adequate nutrition.
- Easing pain and discomfort: Recurrent fevers are common in acute leukemia; at times, they are accompanied by shaking chills (rigors), which can be severe. Myalgias and arthralgias can result.
- Acetaminophen is typically given to decrease fever, but it can also increase diaphoresis.
- Sponging with cool water may be useful, but cold water or ice packs should be avoided because the heat cannot dissipate from constricted blood vessels.
- Bedclothes need frequent changing as well.
- Gentle back and shoulder massage provide comfort.
- Decreasing fatigue and activity intolerance: Fatigue is a common and oppressive symptom. Nursing interventions should focus on assisting the patient to establish a balance between activity and rest. Patients with acute leukemia need to maintain some physical activity and exercise to prevent the deconditioning that results from inactivity.
- The use of a high-efficiency particulate air (HEPA) filter mask can permit the patient to ambulate outside the room despite severe neutropenia.
- Stationary bicycles may also be set up in the room; however, many patients lack the motivation or stamina to use them.
- At a minimum, patients should be encouraged to sit up in a chair while awake rather than staying in bed; even this simple activity can improve the patient’s tidal volume and enhance circulation.
- Physical therapy can also be beneficial.
- Patients with acute leukemia may require hospitalization for extensive nursing care; sleep deprivation frequently results. Nurses need to implement creative strategies that permit uninterrupted sleep for at least a few hours while still administering necessary medications on schedule.
- Maintaining fluid and electrolyte balance: Febrile episodes, bleeding, and inadequate or overly aggressive fluid replacement can alter the patient’s fluid status. Similarly, persistent diarrhea and vomiting that occur with certain chemotherapy and immunotherapy agents, and long-term use of certain antimicrobial agents can cause significant deficits in electrolytes.
- Intake and output need to be measured accurately, and daily weights should also be monitored.
- The patient should be assessed for signs of dehydration and overhydration, with particular attention to pulmonary status and the development of dependent edema.
- Laboratory test results, particularly electrolytes, blood urea nitrogen, creatinine, and hematocrit, should be monitored and compared with previous results.
- Replacement of electrolytes, particularly potassium and magnesium, is commonly required.
- Improving self-care: bathing, dressing, and toileting: Because hygiene measures are so important in this patient population, they must be performed by the nurse when the patient cannot do so. Patients may have negative feelings because they can no longer care for themselves.
- The patient should be encouraged to do as much as possible to preserve mobility and function as well as self-esteem.
- Empathetic listening is helpful, as is realistic reassurance that these deficits are temporary.
- As the patient recovers, the nurse assists them to resume more self-care.
- Patients are usually discharged from the hospital with a vascular access device, and coordination with appropriate home care services is needed for catheter management.
- Managing anxiety and grief: The patient often has little time to process the fact that they have illness before making decisions about therapy.
- Providing emotional support and discussing the uncertain future are crucial.
- The nurse also needs to assess how much information the patient wants to have regarding the illness, its treatment, and potential complications. This desire should be reassessed at intervals, because needs and interest in information change throughout the course of the disease and treatment.
- Priorities must be identified so that procedures, assessments, and self-care expectations are adequately explained even to those who do not wish extensive information.
- Many patients exhibit depressive symptoms and begin to grieve for their losses, such as normal family functioning, professional roles and responsibilities, and social roles, as well as physical functioning. The nurse can assist the patient to identify the source of the grief and encourage them to allow time to adjust to the major life changes produced by the illness.
- Role restructuring, in both family and professional life, may be required. It is essential to encourage the patient to identify options and to take time in making important decisions.
- Providing emotional support to both the patient and the family is critical and equally as important as is rendering expert physical care.
- Discharge from the hospital can also provoke anxiety. Although most patients are eager to go home, they may lack confidence in their ability to manage potential complications and to resume their normal activity. Close communication between nurses across care settings can reassure patients that they will not be abandoned.
- Encouraging spiritual well-being: Because acute leukemia is a serious, potentially life-threatening illness, the nurse may offer support to enhance the patient’s spiritual well-being.
- The patient’s spiritual and religious practices should be assessed and pastoral services offered. Throughout the patient’s illness, the nurse assists the patient to maintain hope.
- However, that hope should be realistic and will certainly change over the course of the illness.
Evaluation
Expected patient outcomes may include:
- Shows no evidence of infection
- Experiences no bleeding
- Has intact oral mucous membranes
- Participates in oral hygiene regimen
- Reports no discomfort in mouth
- Attains optimal level of nutrition
- Maintains weight with increased food and fluid intake
- Maintains adequate protein stores (e.g., albumin, prealbumin)
- Reports satisfaction with pain and comfort levels
- Has less fatigue and increased activity
- Maintains fluid and electrolyte balance
- Participates in self-care
- Copes with anxiety and grief
- Discusses concerns and fears
- Uses stress management strategies appropriately
- Participates in decisions regarding end-of-life care
- Reports sense of spiritual well-being
- Absence of complications
Footnotes
-
A life-threatening complication with symptoms such as unexplained fever, weight gain, hypotension, respiratory distress, and acute kidney injury ↩